September 20, 2018Management
AGC to Establish Mammalian cGMP-Compatible Contract Facility for Biopharmaceutical Development and Production at Chiba Plant
AGC announced today it will be establishing a cGMP*1- compliant mammalian cell culture facility at its Chiba Plant, designed to take on contracts for antibody biopharmaceutical development and production. This is made possible through the technology of CMC Biologics acquired in 2017, and will be the first high-quality mammalian cell culture based services with proven track record in the US and Europe to be made available in Japan. As a large portion of antibody biopharmaceutical products currently depend on overseas production, we look forward to better serving our Japanese customers and the neighboring regions through this new domestic capability. The facility is scheduled to begin operations in the third quarter of 2019.
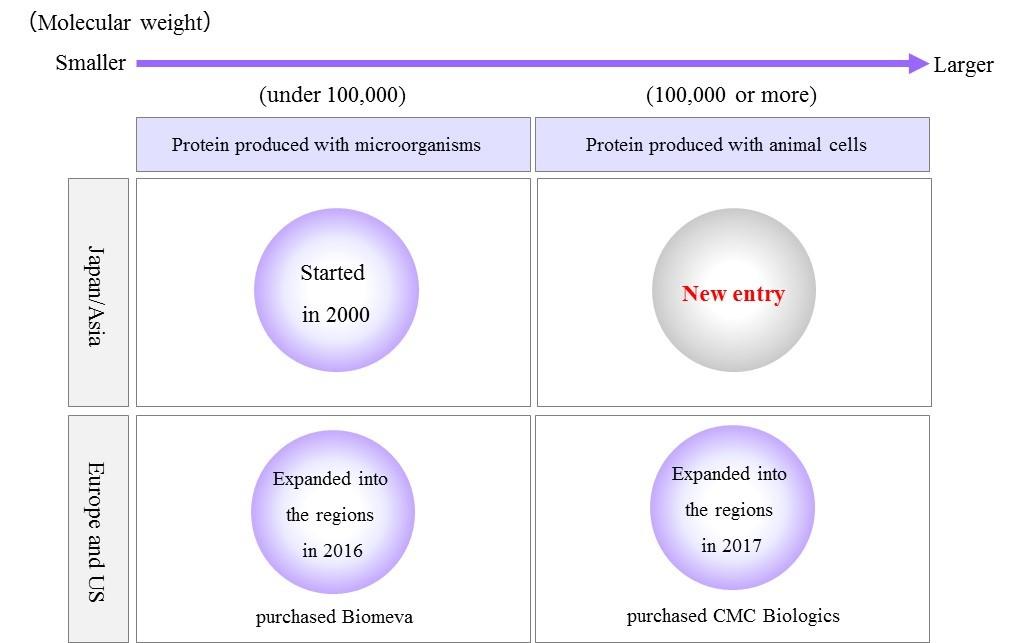
AGC entered the bioscience business in 1985, and launched its biopharmaceutical CDMO*2 business in 2000, with primary focus on the production of microbially-produced protein therapeutic products for the Japanese market. Biomeva was acquired in 2016 and CMC Biologics in 2017, allowing AGC to realize full-scale entry into the Western biopharmaceutical CDMO market, not only using microbial production, but mammalian cells as well. As of January 2018, the biopharmaceutical CDMO businesses at each of the bases in Japan, the US, and Europe have been integrated under a global unified management as AGC Biologics.
Under the management policy AGC plus, the AGC Group will continue to position life sciences as one of its strategic businesses. We will continue to actively invest, so that we can provide globally-unified high-quality services to customers of all regions in the synthetic drug and biopharmaceutical businesses, areas expected to see great growth in demand. In addition, by maximizing the synergies of each location, we will enhance our technical capabilities and contribute to pharmaceutical companies, patients, and wider society.
Notes
*1 cGMP: Production and quality management standards for pharmaceuticals and quasi-drugs (current Good Manufacturing Practice)
*2 CDMO: Contract Development & Manufacturing Organization
*2 CDMO: Contract Development & Manufacturing Organization
References
■About the AGC Group■
AGC Inc.(Headquarters: Tokyo, President & CEO: Takuya Shimamura) is the parent company of the AGC Group, a world-leading glass solution provider and supplier of flat, automotive and display glass, chemicals, ceramics and other high-tech materials and components. Based on more than a century of technical innovation, the AGC Group has developed a wide range of cutting-edge products. The AGC Group employs some 50,000 people worldwide and generates annual sales of approximately 1.5 trillion Japanese yen through business in about 30 countries. For more information, please visit www.agc.com/en
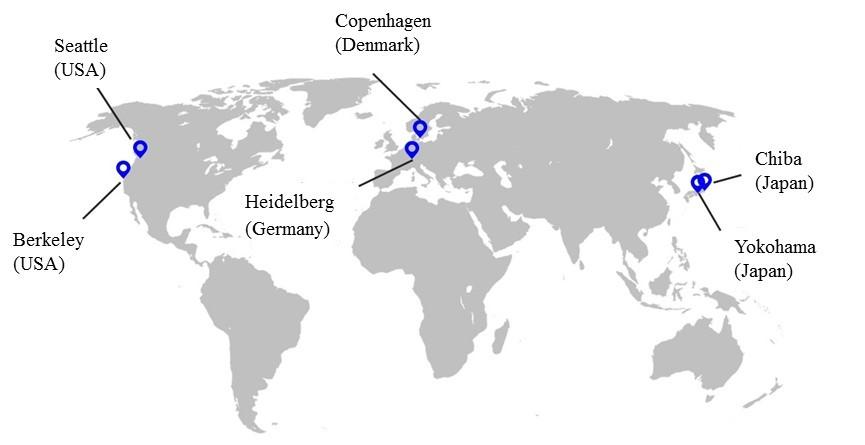
■AGC Group Bioscience Business Locations■
AGC maintains bioscience business bases at the following locations.
AGC Inc.(Headquarters: Tokyo, President & CEO: Takuya Shimamura) is the parent company of the AGC Group, a world-leading glass solution provider and supplier of flat, automotive and display glass, chemicals, ceramics and other high-tech materials and components. Based on more than a century of technical innovation, the AGC Group has developed a wide range of cutting-edge products. The AGC Group employs some 50,000 people worldwide and generates annual sales of approximately 1.5 trillion Japanese yen through business in about 30 countries. For more information, please visit www.agc.com/en
■AGC Group Bioscience Business Locations■
AGC maintains bioscience business bases at the following locations.
■About AGC’s Life Sciences Business■
We will launch a CDMO business using mammalian cells in the Japan/Asia region by establishing a new contract facility for mammalian cell development and production.
We will launch a CDMO business using mammalian cells in the Japan/Asia region by establishing a new contract facility for mammalian cell development and production.
- Media inquiries
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Yuki Kitano
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com