November 11, 2019Management
AGC to Expand US and European Biopharmaceutical Production Facilities
AGC Inc.(AGC), a world-leading manufacturer of glass, chemicals and high-tech materials, has announced its decision to expand facilities at the US and European sites of its subsidiary AGC Biologics, headquartered in the United States and engaged in the biopharmaceutical CDMO*1 business.
A 500-liter mammalian cell bioreactor will be added to its Seattle facility in the US, while a new purification line will be installed at its Copenhagen facility in Europe. The total investment is expected to be around 2 billion yen. Operation is scheduled to begin at the US facility in April 2020, and October at the European facility.
 AGC Biologics, Seattle Facility
AGC Biologics, Seattle Facility
AGC Group has made a commitment to positioning life sciences, which includes its biopharmaceutical CDMO business, as one of its strategic initiatives with the aim of achieving sales in the 100-billion-yen range by 2025. Through this initiative, AGC has already developed manufacturing and sales bases in Europe and the US through the acquisitions of Biomeva (Germany) in 2016 and CMC Biologics (United States and Denmark) in 2017. Since then, the company has been making aggressive investments in equipment and facilities at locations across Japan, Europe, and the US, which has succeeded in steadily expanding contracted work.
A 500 liter single-use*2 mammalian cell bioreactor will be added to the company's Seattle facility in the US. This will serve better, pharmaceuticals companies with biopharmaceuticals at the earlier stages of development and clinical trials. Meanwhile, a new purification line will be installed at the company's Copenhagen facility in Europe, along with additional analytic labs. This will boost the Copenhagen site’s production efficiency by 1.5 times. These expansions will help meet the growing market, which has been growing at a pace of at least 8% per year.
Under its AGC plus management policy, the AGC Group intends to continue aggressively pursuing necessary acquisitions and capital investment in life sciences business, expected to exhibit significant growth in demand in the coming years, in order to continue providing customers in every region with globally consistent, top-level quality and service. By maximizing synergies among operation sites, AGC will continue to pursue its goal of being a leading company that supports pharmaceutical companies, medical patients, and all of society.
Notes:
*1 CDMO: Contract Development & Manufacturing Organization. A company which is contracted or acts on behalf of another company to handle product manufacturing as well as the development of manufacturing methods.
*2 Single-use: Culture tanks that use a disposable vessel
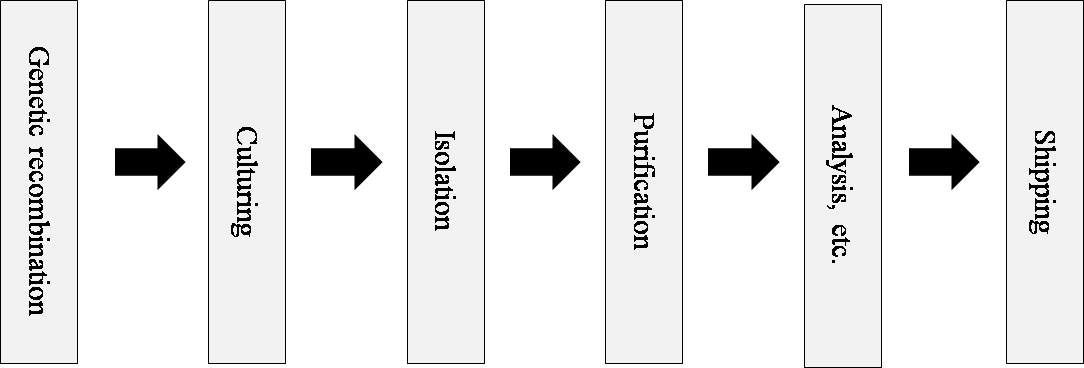
■ Biopharmaceutical CDMO Process
The biopharmaceutical CDMO manufacturing process is as shown below. The facility enhancements will facilitate culturing processes at the Seattle facility and purification processes at the Copenhagen facility.
The biopharmaceutical CDMO manufacturing process is as shown below. The facility enhancements will facilitate culturing processes at the Seattle facility and purification processes at the Copenhagen facility.
- MEDIA INQUIRIES
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Yuki Kitano
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com