April 7, 2020Management
AGC to Expand its Spanish Synthetic Pharmaceutical Production Base
Increase of Production Capacity and Establishment of a New R&D Facility
This initiative will not only upgrade existing production facilities to achieve 1.3 times the capacity of the current production level, but also establish a brand-new R&D facility. The new R&D facility is scheduled to begin operation in March 2021, while the expanded facility is scheduled to begin in May 2022.
The synthetic pharmaceutical CDMO market is growing at over 7% annually, while the number of contracts handled by the company are growing at an even faster rate. To satisfy this accelerating demand, existing production facilities will be upgraded to increase pharmaceutical production capacity by 1.3 times that of the current level. A new micronization facility will also be introduced to respond to the increasing demand for micronization of active pharmaceutical ingredients. In addition, a brand-new R&D facility will be built which will enable development of processes from lab scale to commercial production, and to deliver better and faster services to customers.
Notes:
*1 Synthetic pharmaceuticals: Pharmaceuticals manufactured through chemical synthesis
*2 CDMO: Contract Development & Manufacturing Organization. A company which is contracted or acts on behalf of another company to handle product manufacturing as well as the development of manufacturing methods.
*3 GMP: (Good Manufacturing Practice) a standard for the manufacture and quality management of pharmaceuticals and quasi-pharmaceutical products.
REFERENCE
| Company name: | AGC Pharma Chemicals Europe S.L.U. |
| Established | 2018 |
| Address | Catalonia, Spain |
| Shareholding ratio | AGC, 100% |
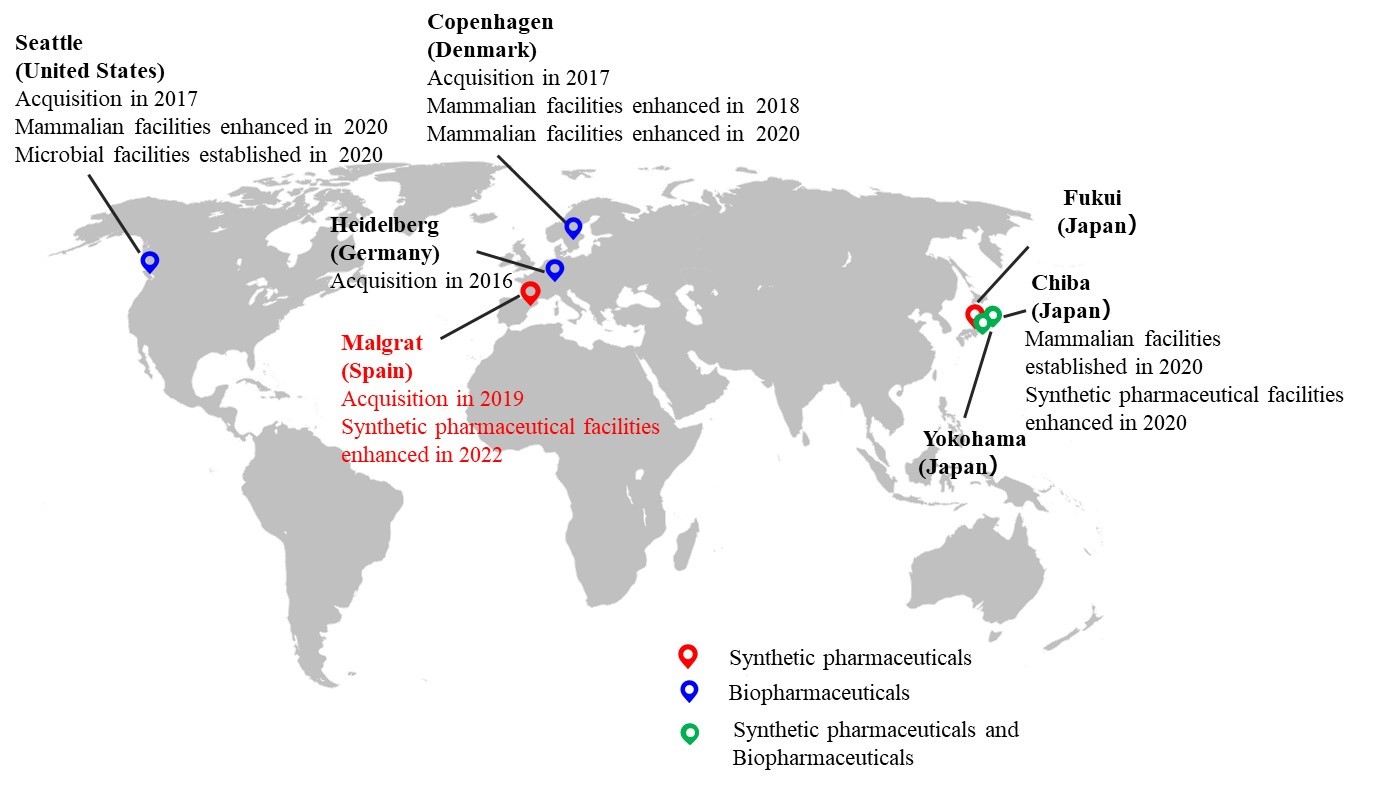
■ AGC Group offices engaged in life-science operations
- MEDIA INQUIRIES
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Yuki Kitano
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com