June 2, 2020Management
AGC Acquires Biopharmaceutical Commercial Facility in Colorado, U.S.A.
AGC a world-leading manufacturer of glass, chemicals and high-tech materials, announced the purchase of a biopharmaceutical commercial manufacturing facility in Boulder, Colorado, USA. The facility, formerly owned by AstreaZeneca, will provide AGC with additional capacity and significantly larger production scale. AGC will prepare for the plant to resume full-scale operations by April 2021.
 Colorado plant exterior
Colorado plant exterior
The Colorado facility is a large-scale biopharmaceutical manufacturing plant that houses two 20,000-liter (total volume) stainless steel mammalian cell bioreactors, and is well suited for higher volume commercial production. It also has more than twenty acres of land providing opportunities for future expansions. The existing plant has space for up to four more 20,000-liter bioreactors. With its expanded customer portfolio, as well as current projects advancing into commercial phase, AGC Biologics was looking to expand its footprint. This addition will enable AGC Biologics to support a wider range of commercial demand, and fulfill the needs of the pharmaceutical industry better.
Notes:
1) CDMO: Contract Development & Manufacturing Organization. A company which is contracted or acts on behalf of another company to handle product manufacturing as well as the development of manufacturing methods.
2) Single-Use: Bioreactors that use a disposable vessel
REFERENCE
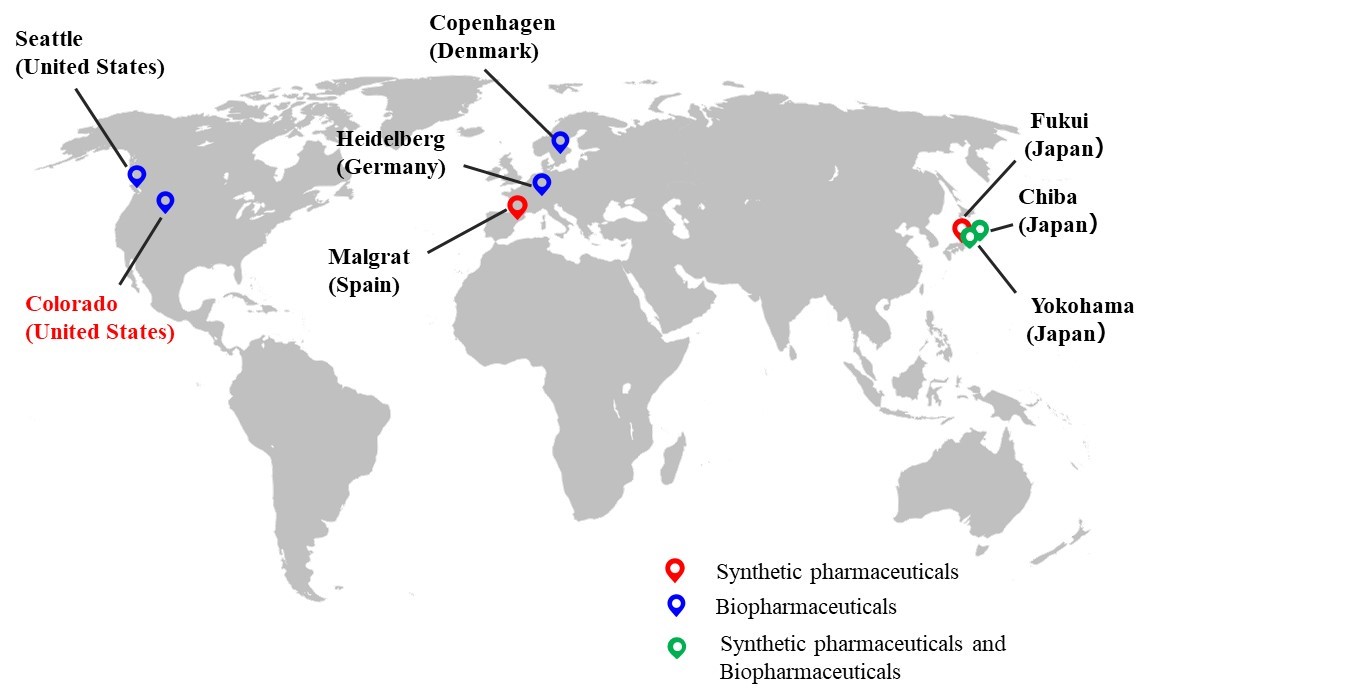
■ AGC Group offices engaged in life-science operations
- MEDIA INQUIRIES
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Yuki Kitano
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com