July 28, 2020Management
Definitive results of the totalitarian voluntary public tender offer promoted by AGC Biologics Italy S.p.A. on all ordinary shares of MolMed S.p.A.
Capitalized terms in this press release, unless otherwise defined, have the meaning attributed to them in the offer document approved by Consob with resolution no. 21391 of 29 May 2020 and published on the same date (the "Offer Document").
1. Fulfilment of the MAC Condition
The MAC Condition referred to in Section A, Paragraph A.1. (letter c)) of the Offer Document shall be considered fulfilled and, therefore, since the Golden Power Condition has already been waived by the Offeror with the press release disseminated on July 10, 2020 and the Threshold Condition has already been fulfilled, the Offer shall be considered fully effective.
2. Provisional results of the Offer
On the basis of the definitive results of the Offer communicated by Banca IMI S.p.A. – the intermediary in charge of coordinating the collection of acceptances to the Offer – during the Acceptance Period no. 432,083,197 Shares, equal to approximately 93,232% of the Shares subject to the Offer and, therefore, to approximately 93,232% of the Issuer's share capital, have been tendered to the Offer, having a total value (calculated on the basis of the Price) equal to EUR 223,819,096.04. Therefore, the total quantity of Shares tendered to the Offer during the Acceptance Period is higher by no. 1,600 Shares compared to the provisional results that was disclosed on behalf of the Offeror on July 24, 2020.
It should be noted that, in the period between the Offer Document Date and the date of this press release, neither the Offeror nor the Persons Acting in Concert have made, directly or indirectly, any purchase of Shares on the market outside the Offer.
Consequently, taking into account the no. 432,083,197 Shares tendered to the Offer, at completion of the Offer the Offeror will hold a shareholding equal to 93,232% of the Issuer's share capital. The Offeror will therefore purchase all the Shares tendered to the Offer during the Acceptance Period and the Price due by the Offeror for each Share tendered during the Acceptance Period, equal to EUR 0.518, will be paid to the shareholders adhering to the Offer on Friday July 31, 2020, against the simultaneous transfer of the right of ownership on such Shares in favor of the Offeror.
By means of a press release to be issued by the Payment Date – i.e. by 31 July 2020 – the Offeror will also disclose terms and timing according to which the Offeror will fulfil the Obligation to Acquire pursuant to Article 108, second paragraph, of the CLFI and on the timing of the subsequent Delisting of the Issuer's Shares from the MTA.
Originating from its own drug discovery efforts, MolMed brings deep experience and expertise in providing development and GMP*2 manufacturing services for viral vectors*3 and genetically modified cells, from the preclinical phase through commercial demand.
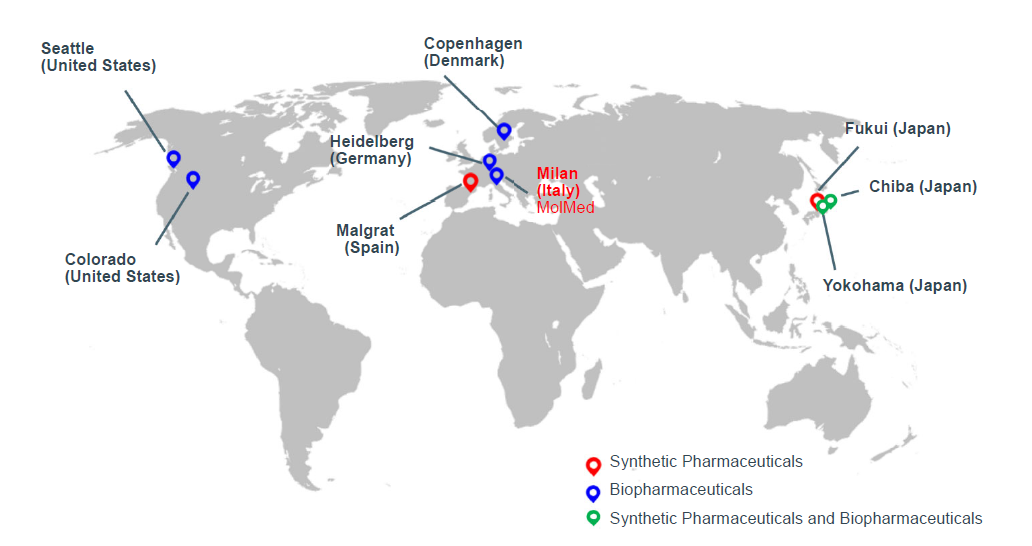
Operating in the three regions of Japan, the US, and Europe, AGC Biologics offers deep industry expertise and unique customized CDMO services for the scale-up and cGMP manufacture of protein-based therapeutics, from pre-clinical to commercial mammalian and microbial production. As the company has already commercialized plasmid production at its Heidelberg Site in Germany, AGC Biologics will now be one of the very few CDMOs offering both plasmid production and cell and gene therapy services.
 MolMed Milano sites
MolMed Milano sites
Notes:
*1 CDMO: Contract Development & Manufacturing Organization. A company which is contracted or acts on behalf of another company to handle product manufacturing as well as the development of manufacturing processes.
*2 GMP: Good Manufacturing Practices. A standard for the manufacture and quality management of pharmaceuticals and quasi-pharmaceutical products.
*3 Vector: A vehicle used to carry the target gene into a cell
REFERENCE
| Established | 1996 |
| Location | Milan, Italy |
| Number of employees | Approx. 220 |
| Sales | 36.3 million euros (2019) |
| Business activities | Gene/cell therapy drug discovery and CDMO |
| Total shares issued | 463,450,672 shares |
| Website | https://www.molmed.com/en/node/1 |
■AGC Group's CDMO Business Sites
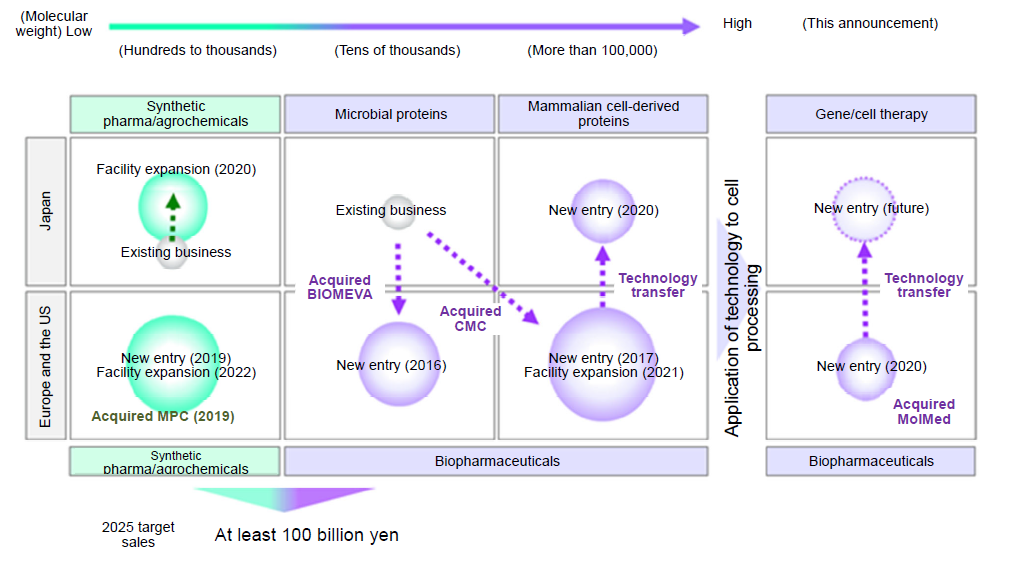
■Where this acquisition fits in the Company's life sciences business
- MEDIA INQUIRIES
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Yuki Kitano
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com