July 2, 2021Management
AGC Biologics Enters Agreement to Acquire Facility in Longmont, Colorado, U.S.
―Eyeing to Significantly Expand Their Cell and Gene Therapy Capabilities and Offerings―
AGC (AGC Inc.; Headquarters: Tokyo; President: Yoshinori Hirai), a world-leading manufacturer of glass, chemicals, and high-tech materials, has announced that its biopharmaceutical CDMO*1 subsidiary, AGC Biologics (Headquarters: United States), has signed an agreement with Novartis Gene Therapies (Headquarters: United States) to purchase a cell and gene therapy manufacturing facility located in Longmont, Colorado, U.S. The closing of the transaction is pending completion of final due diligence.
The addition of this facility follows last year’s acquisition of the Italian company MolMed (now AGC Biologics S.p.A., referred to as “Milan Site”), which is currently undergoing expansion previously announced. With the needs of customers rapidly increasing in the fast-growing field of gene and cell therapy, AGC Biologics is securing further manufacturing capacity, this time in the United States, the world-leading market in this field. Once the transaction closes, the facility will be integrated into AGC Biologics' network, and services will be extended incorporating the Milan Site’s robust experience providing commercial GMP*2-compliant cell and gene therapy CDMO services, utilizing the facility’s large floor space at over 60,000 square meters. Further, the Heidelberg Site has an established plasmid DNA production service, enabling AGC Biologics to offer end-to-end cell and gene services. This facility will become part of that offering.
 The facility to be purchased
The facility to be purchased
Under its AGC plus 2.0 management policy, AGC Group has positioned its Life Science business including its biopharmaceutical CDMO services, as one of its strategic businesses. The Group has been expanding its business through multiple acquisitions and capital investments for its CDMO services using mammalian cells and microorganisms, as well as in synthetic pharmaceutical and agrochemical services. Furthermore in 2020, the Group expanded the scope of its CDMO business into the rapidly growing field of cell and gene therapy. The sales target for the Group’s Life Science business is 180 billion yen or more for 2025.
The AGC Group will work to provide its customers in each region with globally unified, high-quality services, contributing to the well-being of patients and the wider society as a whole.
Notes:
*1 CDMO: Contract Development & Manufacturing Organization. A company which is contracted on behalf of another company to provide product manufacturing services as well as the development of manufacturing processes.
*2 GMP: Good Manufacturing Practice. A standard for the manufacture and quality management of pharmaceutical products.
REFERENCE
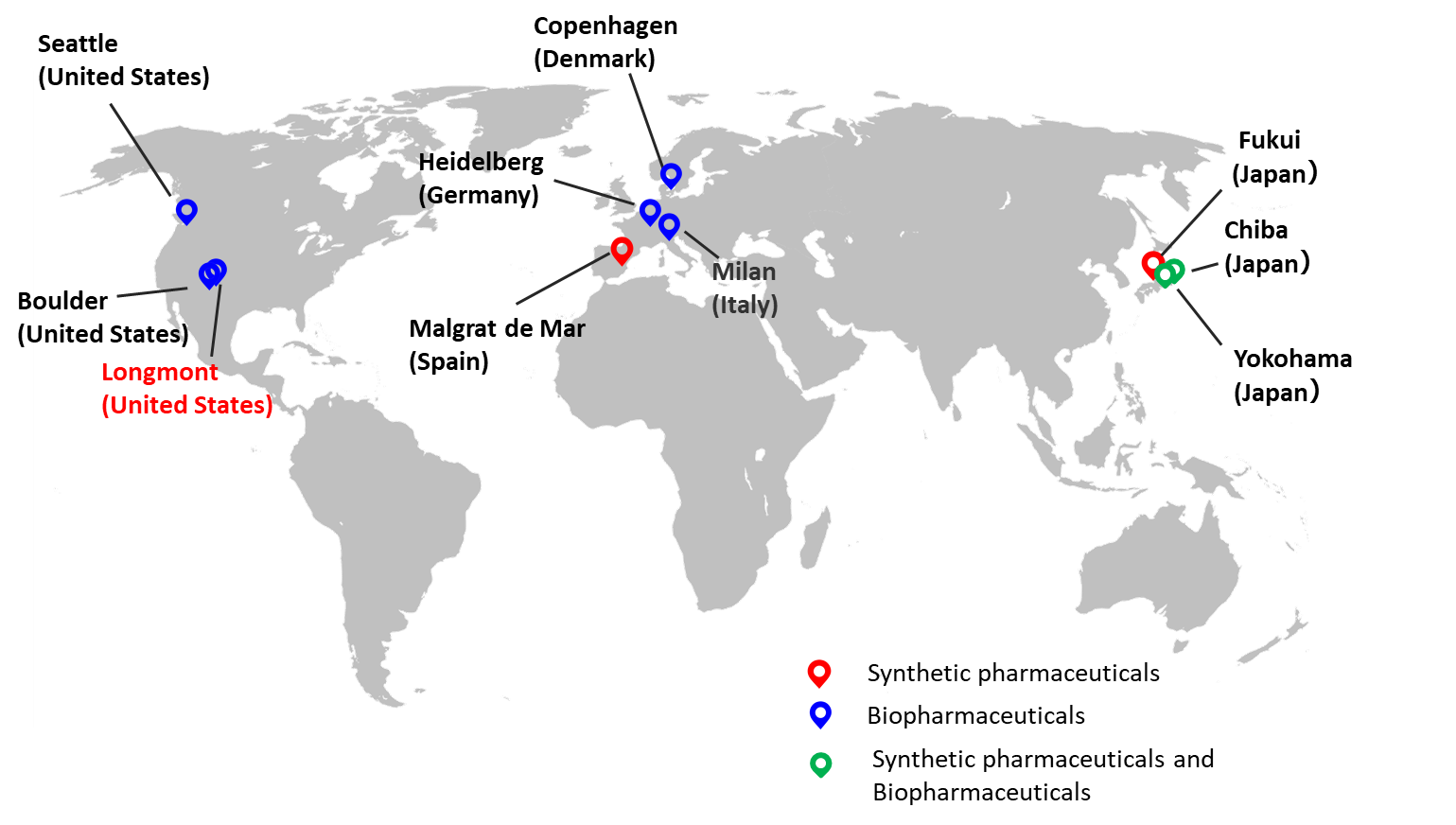
■AGC Group’s CDMO Business Sites
- MEDIA INQUIRIES
- Kazumi Tamaki, General Manager, Corporate Communications & Investor Relations Division
AGC Inc. - Contact: Tomoko Nakao
- TEL: +81-3-3218-5603
- E-mail: info-pr@agc.com